- Pack/Packs
- 500
- Pack/Packs
- 1.00 - 5.00 USD ($)
- 1.5 USD ($)
- External Use Drugs
- Capsules
- Ocular hypertension, Chronic open-angle glaucoma, Some cases of secondary glaucoma
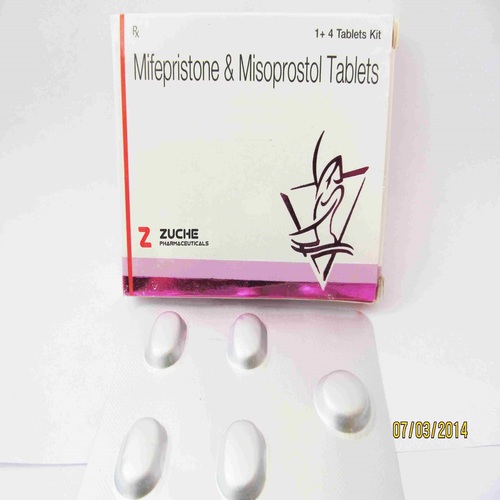
Product Name: Mifepristone and Misoprostol Tablets
Common Name: Mifepristone and Misoprostol Tablets
Description:
Medical Abortion is a form of early abortion caused by the combination of two medications, mifepristone and misoprostol that is an option for women who are 8 weeks pregnant or less. Also known as RU486 or medication abortion.
During the first appointment at the clinic you receive the mifepristone pill to take orally. Then 24 to 72 hours later, in the privacy, take the the second medication, misoprostol. Misoprostol causes contractions resulting in a miscarriage. When used in combination, mifepristone and misoprostol are 95-97% effective within two weeks. Mifepristone and misoprostol are FDA approved.
Indications and Usage:
Mifepristone is used in combination with misoprostol (Cytotec) to end an early pregnancy. Early pregnancy means it has been 70 days or less since your last menstrual period began. Mifepristone is in a class of medications called antiprogestational steroids. It works by blocking the activity of progesterone, a substance your body makes to help continue pregnancy.
Mifepristone is also available as another product (Korlym), which is used to control hyperglycemia (high blood sugar) in people with a certain type of Cushing's Syndrome in which the body makes too much of the hormone cortisol. This monograph only gives information about mifepristone (Mifeprex), which is used alone or in combination with another medication to end an early pregnancy.
Mifepristone is also sometimes used to end pregnancies when more than 70 days have passed since the woman's last menstrual period; as an emergency contraceptive after unprotected sexual intercourse ('morning-after pill'); to treat tumors of the brain, endometriosis (growth of uterus tissue outside the uterus), or fibroids (noncancerous tumors in the uterus); or to induce labor (to help start the birth process in a pregnant woman).
Features:
1) WHO GMP Approved Plant
2) Customized Packing Available
3) Regulatory and documentation support available
Pack Size: Each pack has 4 Tablets
Minimum Order Quantity: 100000 tablets
- New Delhi
- 100000 Per Month
- 30 Days
- Yes
- Sample costs shipping and taxes has to be paid by the buyer
- Each pack has 4 Tablets
- WHO GMP Approved Plant
109, first floor, Oriental House 19-20, Community Centre Complex, Yusuf Sarai, Gulmohar Park, New Delhi, Delhi, 110049, India
Phone :+918045479428